Evaluating Exosome-Based Therapies: A Clinical Framework for Practitioners
- Yingxuan Lim
- 1 hour ago
- 3 min read
Part 2 of 3 — Bridging the Exosome Gap · For Clinicians & Practitioners
Missed Part 1? Read: Why Autologous Exosomes Outperform Non-Autologous Exosomes

The exosome therapy market is expanding rapidly, and with it comes an equally rapid proliferation of commercial products — many of which make claims that outpace the evidence. As a clinician, you are already fielding questions from patients who have encountered exosome products marketed across aesthetics, regenerative medicine, and hair restoration.
The challenge is not whether exosome biology is sound — it is. The challenge is distinguishing the therapies that are clinically defensible from those that expose you and your patients to unnecessary risk. This article provides the five-question framework we recommend every practitioner apply when evaluating any exosome-based product or therapy.
Each question addresses a specific domain where the gap between marketing claims and clinical reality is widest — and where MCT Exosomes offer a substantively different position.
Commercial exosome products are routinely marketed with headline particle counts — five billion, ten billion, or more exosomes per vial — positioned as a proxy for therapeutic potency. This framing does not reflect current scientific understanding of exosome dose-response relationships.



The concentration figures provided on commercially available exosome vials are derived from biophysical measurement methods that share a fundamental limitation: none can selectively enumerate exosomes to the exclusion of co-isolating particles.

Compounding the measurement problem is the absence of a universally accepted quantification standard. Different manufacturers use different methods, reference materials, and protocols — making cross-product comparisons unreliable. Additionally, particles that routinely co-isolate with exosomes include microvesicles (100–1000 nm), retroviruses (80–150 nm), exomeres (~30–50 nm), HDL/LDL lipoproteins (5–35 nm), and apoptotic vesicles — all of which may be counted as part of the reported exosome concentration.



This is the question with the most direct implications for practitioner liability. The majority of exosome products currently available on the market are classified as cosmetics — a regulatory category that expressly prohibits administration by injection. Understanding this distinction is not optional for practitioners.
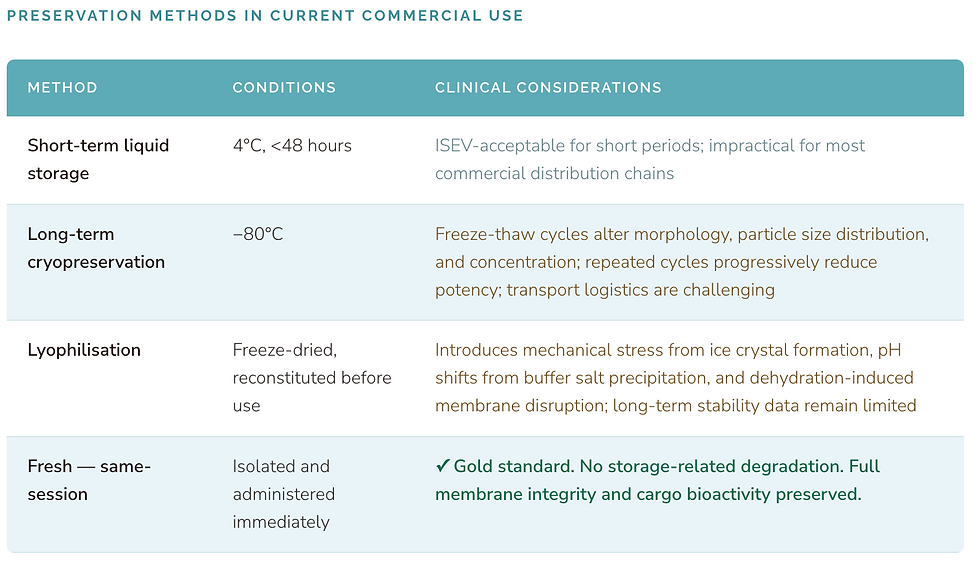
Research by Frank et al. on EV lyophilisation demonstrated reduced particle counts relative to 4°C and −80°C storage conditions, indicating particle loss or aggregation during the freeze-drying process. Among cryoprotectants evaluated, trehalose demonstrated superiority to mannitol and polyethylene glycol 400, though comprehensive long-term stability data for lyophilised EV preparations remain lacking. The consensus position of the field is consistent: freshly obtained extracellular vesicles outperform stored preparation


This is the question with the most direct implications for practitioner liability. The majority of exosome products currently available on the market are classified as cosmetics — a regulatory category that expressly prohibits administration by injection. Understanding this distinction is not optional for practitioners.


For exosomes to exert their regenerative effects, internalisation by metabolically active target cells via endocytosis is required. Cells in the stratum corneum — the primary contact layer for topically applied products — are anucleate and lack the biological machinery for vesicle internalisation. The clinical case for topical exosome application as a regenerative intervention is therefore limited by basic cell biology, independent of the regulatory classification question.


A recurring claim in exosome product marketing is the concept of tissue-specific targeting — that a given preparation can be directed to hair follicles, a specific skin layer, or a particular organ. The current scientific evidence for intrinsic exosome tropism in healthy-cell-derived preparations does not support these claims at the level of specificity implied.

Unlike viruses, which have evolved highly specific receptor interactions enabling selective tissue infection, exosomes from healthy parental cells do not carry equivalent "address labels." While surface molecules including tetraspanins (CD63, CD9, CD81) and integrins have been proposed as potential determinants of preferential biodistribution, this phenomenon has been characterised primarily in cancer cell-derived EVs and apoptotic MSC vesicles — not in naïve exosomes from healthy donor cells. Some studies have further demonstrated that EVs do not reliably traffic in a targeted manner in vivo, raising questions about their utility as autonomous drug delivery vehicles independent of administration site.

The Five-Question Clinical Evaluation Framework
When any exosome-based therapy is presented to your clinic — whether by a supplier, a distributor, or an enquiring patient — these are the questions that determine whether it meets the standard of a clinically and regulatorily defensible intervention:

MCT Exosomes provide a clinically and regulatorily substantiated answer to each of these questions. Autologous origin, same-session freshness, certified injectable classification, and direct delivery to the treatment site are the foundations of a therapy you can stand behind with your patients and within your regulatory obligations.
Ready to Experience the MCT Difference?
Speak with the Athena Biomed team to book a demo today.
Join the future of regeneration.
.png)

