5 Reasons Autologous Exosomes Outperform Non-Autologous Exosomes
- IFAAS Ops
- Apr 14
- 4 min read
Updated: 1 day ago
Introduction
Building on the foundation of The Power of Injectable Autologous Exosomes: Introducing the MCT Metacell Technology System, it becomes clear that the true differentiator in exosome-based therapy is not merely the presence of exosomes—but the origin, integrity, and method of delivery.
The MCT framework is specifically engineered around one core principle: that clinically effective regenerative signaling must be both biologically compatible and precisely delivered. This is where the distinction between autologous and non-autologous exosomes becomes critically important.
From Technology to Biology: Why Source Defines Outcome
While the MCT system enables the extraction, concentration, and injection of high-potency exosomes, its clinical advantage is fundamentally rooted in its exclusive use of autologous material.
Exosomes can be broadly categorized into:
Autologous (patient-derived)
Allogeneic (donor-derived, same species)
Xenogeneic (cross-species, including animal or plant sources)
The MCT system deliberately avoids non-autologous sources—not as a limitation, but as a clinical necessity.
1. Immune compatibility focus: One of the defining strengths of MCT—fully autologous injectable exosomes—directly addresses the most immediate risk in regenerative therapy: immune incompatibility.
Because MCT-derived exosomes originate from the patient:
They retain native surface markers recognized as “self.”
They integrate seamlessly into local cellular environments
They avoid immune activation entirely
This aligns precisely with MCT’s positioning as a safe, injectable platform, where introducing foreign biological material would undermine both efficacy and patient safety.
In contrast, non-autologous exosomes inherently contradict this principle:
They carry foreign MHC molecules and antigens
They may trigger inflammatory or immune-mediated responses
They are often rapidly cleared, reducing therapeutic persistence
Thus, the injectable nature of MCT amplifies the importance of autologous sourcing—what may be tolerated topically becomes clinically unacceptable when delivered into tissue.

2. Higher potency precision
A central claim of MCT is its superior safety profile, and this is inseparable from its autologous design.
Non-autologous exosomes introduce variables that are incompatible with high-standard medical practice:
Risk of viral, bacterial, or cross-species contamination
Transfer of foreign proteins or pathogenic signals
Theoretical risks related to prion-associated or neurodegenerative pathways
MCT eliminates these risks at the source level.
By maintaining a closed, patient-specific biological loop, the system ensures:
No donor dependency
No cross-contamination
No unknown biological exposure
This is particularly critical for clinicians seeking predictable, reproducible outcomes in injectable therapies. 3. Greater source standardization
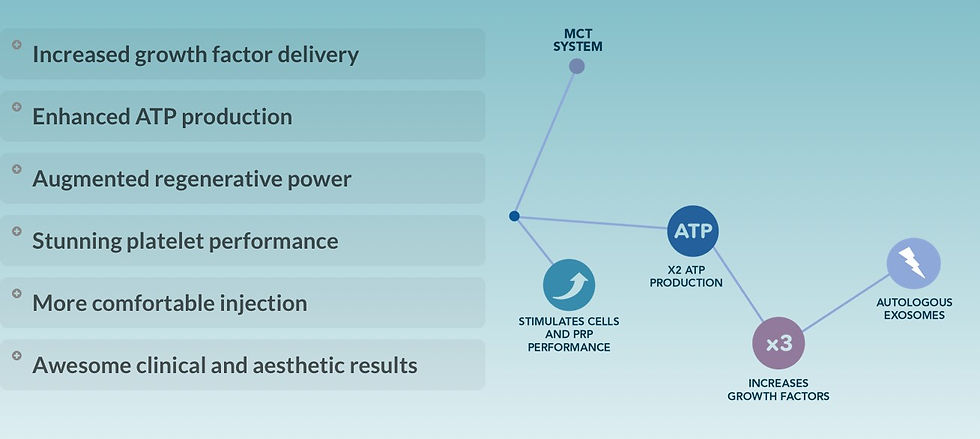
MCT’s ability to deliver ultra-high concentrations (up to 300 billion exosomes per mL) is only clinically meaningful if those exosomes are functionally appropriate and biologically coherent.
Autologous sourcing ensures that:
The exosome cargo reflects the patient’s physiological state
Signaling pathways remain relevant and targeted
Cellular responses are aligned with the intended regenerative outcome
In contrast, high concentrations of non-autologous exosomes may:
Amplify unwanted or nonspecific signals
Increase the risk of off-target effects
Deliver biologically incompatible instructions
Therefore, potency without compatibility is not an advantage. MCT’s strength lies in combining both high signal density and biological precision.
4. Systemized therapeutic design
MCT is often compared to PRP, particularly in its ability to deliver 3× higher growth factor activity. However, the distinction goes beyond quantity—it is fundamentally about how those signals are delivered and interpreted.
PRP relies on:
Passive release of growth factors
Variable activation depending on preparation and patient condition
MCT, through autologous exosomes, provides:
Encapsulated, protected signaling molecules
Targeted intercellular communication via vesicle-mediated delivery
Sustained and regulated biological effects
This makes source integrity even more critical. Autologous exosomes ensure that these advanced delivery mechanisms remain:
Safe
Predictable
Clinically meaningful
5. More consistent safety profile Many exosome products on the market emphasize:
“Younger” donor sources (e.g., umbilical cord, placental, animal-derived)
Convenience through off-the-shelf availability
However, when evaluated through the MCT standard, these claims fall short:
They remain non-autologous, and therefore immunologically foreign
They introduce cargo uncertainty, undermining precision medicine
They are often non-injectable, limiting their clinical utility
MCT redefines the benchmark by prioritizing:
Biological compatibility over marketing narratives
Clinical safety over convenience
Injectable precision over superficial application
Integrated Perspective: Why MCT and Autologous Exosomes Are Inseparable
The advantages of autologous exosomes are not separate from MCT—they are fully enabled and expressed through the system itself.
MCT provides a controlled, standardized platform to extract high-quality autologous exosomes while preserving their biological integrity for clinical use. It then concentrates these exosomes to therapeutically relevant levels, converting a naturally low-abundance signaling system into a potent regenerative dose. Finally, it supports precise delivery to targeted tissues, ensuring that regenerative signals are administered exactly where they are required.
At the same time, autologous sourcing ensures the system operates within a safe immunological framework, as the exosomes are inherently recognized as “self,” eliminating risks of immune activation or rejection. This also creates a predictable biological profile, with signaling behavior aligned to the patient’s native physiology rather than donor variability. Together, this enables MCT to function as a fully patient-specific regenerative platform, where safety, consistency, and efficacy are inherently aligned.
Conclusion

The concept of “The Power of Injectable Autologous Exosomes” is not theoretical—it is operationalized through the MCT Metacell Technology System.
By integrating autologous sourcing for safety and biocompatibility, injectable delivery for precise tissue targeting, and ultra-high potency for clinically meaningful effects, MCT establishes a unified regenerative platform.
In this framework, source, system, and therapeutic outcome are tightly aligned. The superiority of autologous exosomes is therefore not merely a scientific consideration, but the foundational principle that defines MCT’s clinical value and performance. Reference:
Alberro, A., Sáenz-Cuesta, M., Muñoz-Culla, M., Mateo-Abad, M., Gonzalez, E., Pérez-Gonzalez, R., … Olascoaga, J. (2014). Increased circulating exosomes in healthy aging and in neurodegenerative conditions. Journal of Extracellular Vesicles, 3(1), 24238. https://doi.org/10.3402/jev.v3.24238
Cui, G.-H., Wu, J., Mou, F.-F., Xie, W.-H., Wang, F.-B., Wang, Q.-G., & Fang, L.-Q. (2018). Exosomes derived from mesenchymal stem cells facilitate cutaneous wound healing via enhancing angiogenesis. Journal of Cellular and Molecular Medicine, 22(10), 4869–4881. https://doi.org/10.1111/jcmm.13774
Elahi, K. C., Klein, G., Avci-Adali, M., Sievert, K. D., MacNeil, S., Aicher, W. K., & Stamp, L. (2021). Exosomes of mesenchymal stem cells: An emerging therapeutic resource for wound healing and tissue regeneration. Stem Cell Research & Therapy, 12, 1–15. https://doi.org/10.1186/s13287-021-02300-5
Discover Autologous Exosomes:
The Future of Aesthetic & Regenerative Medicine is Here
Join the free Athena Biomed Webinar

22 April 2026 (Wed) | [Register Now]
Discover MCT Metacell with Athena Biomed
.png)
